There are 2 main 'bases' for engine coolant Ethylene Glycol and Propylene Glycol.
Ethylene Glycol:-
Ethylene glycol solutions became available in 1926 and were marketed as "permanent antifreeze," since the higher boiling points provided advantages for summertime use as well as during cold weather. They are used today for a variety of applications, including automobiles. Ethylene glycol has been ingested on occasion, causing ethylene glycol poisoning.
Ethylene glycol antifreeze has a sweet taste that can contribute to its accidental ingestion or its deliberate use as a murder weapon, as attributed by sensational media reports concerning it. Symptoms of antifreeze poisoning include severe diarrhea and vomiting. Farther into the intoxication, signs of delirium, paranoia and intense hallucinations manifest. Antifreeze poisoning can be identified from the growth of calcium oxalate crystals in the kidneys. An embittering agent such as denatonium can be added to ethylene glycol to help discourage either accidental or deliberate poisoning.
Ethylene glycol is toxic to many animals, including cats and dogs, so waste antifreeze should be disposed properly.
Propylene Glycol:-
Propylene glycol, on the other hand, is considerably less toxic and may be labeled as "non-toxic antifreeze." It is used as antifreeze where ethylene glycol would be inappropriate, such as in food-processing systems or in water pipes in homes where incidental ingestion may be possible.
Propylene glycol oxidizes when exposed to air and heat. When this occurs lactic acid is formed. If not properly inhibited, this fluid can be very corrosive. pH buffering agents are added to propylene glycol, preventing low pH attack on the system metals.
Besides cooling system breakdown, biological fouling also occurs. Once bacterial slime starts, the corrosion rate of the system increases. Maintenance of systems using glycol solution includes regular monitoring of freeze protection, pH, specific gravity, inhibitor level, color, and biological contamination.
(*information taken from [ame="http://en.wikipedia.org/wiki/Antifreeze"]Antifreeze - Wikipedia, the free encyclopedia@@AMEPARAM@@/wiki/File:Ethylene_glycol_chemical_structure.png" class="image"><img alt="" src="http://upload.wikimedia.org/wikipedia/commons/thumb/2/28/Ethylene_glycol_chemical_structure.png/100px-Ethylene_glycol_chemical_structure.png"@@AMEPARAM@@commons/thumb/2/28/Ethylene_glycol_chemical_structure.png/100px-Ethylene_glycol_chemical_structure.png[/ame]*)
It's advisable to use Distilled water when changing coolant as tap water will create 'furring' in the cooling system, possibly blocking up smaller coolant galleries over time.
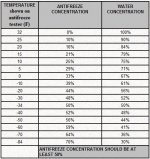
Concentrations vary depending on manufacturer and application but are generally between a 40%-60% mix. Altho Some coolants for colder climates may have a stronger mix of 70%-30% which can offer protection down to -64*C (-84*F) Make sure you check the bottle to ensure correct concentration for that particular coolant in your climate.
this is a rough guide to coolant concentrates and should be used for information only every coolant differs so make sure to read the bottle before starting.
*I cannot guarantee the accuracy of the information in this document nor do I take any responsibility for damage to any vehicle or person due to the misinterpretation of the information/inadequate skills, any work that you carry out to your own vehicle is entirely at your own risk. These are guidelines only and if in doubt I kindly suggest that you contact a Fiat dealer/relative organisation for clarification/legalities.*
Ethylene Glycol:-
Ethylene glycol solutions became available in 1926 and were marketed as "permanent antifreeze," since the higher boiling points provided advantages for summertime use as well as during cold weather. They are used today for a variety of applications, including automobiles. Ethylene glycol has been ingested on occasion, causing ethylene glycol poisoning.
Ethylene glycol antifreeze has a sweet taste that can contribute to its accidental ingestion or its deliberate use as a murder weapon, as attributed by sensational media reports concerning it. Symptoms of antifreeze poisoning include severe diarrhea and vomiting. Farther into the intoxication, signs of delirium, paranoia and intense hallucinations manifest. Antifreeze poisoning can be identified from the growth of calcium oxalate crystals in the kidneys. An embittering agent such as denatonium can be added to ethylene glycol to help discourage either accidental or deliberate poisoning.
Ethylene glycol is toxic to many animals, including cats and dogs, so waste antifreeze should be disposed properly.
Propylene Glycol:-
Propylene glycol, on the other hand, is considerably less toxic and may be labeled as "non-toxic antifreeze." It is used as antifreeze where ethylene glycol would be inappropriate, such as in food-processing systems or in water pipes in homes where incidental ingestion may be possible.
Propylene glycol oxidizes when exposed to air and heat. When this occurs lactic acid is formed. If not properly inhibited, this fluid can be very corrosive. pH buffering agents are added to propylene glycol, preventing low pH attack on the system metals.
Besides cooling system breakdown, biological fouling also occurs. Once bacterial slime starts, the corrosion rate of the system increases. Maintenance of systems using glycol solution includes regular monitoring of freeze protection, pH, specific gravity, inhibitor level, color, and biological contamination.
(*information taken from [ame="http://en.wikipedia.org/wiki/Antifreeze"]Antifreeze - Wikipedia, the free encyclopedia@@AMEPARAM@@/wiki/File:Ethylene_glycol_chemical_structure.png" class="image"><img alt="" src="http://upload.wikimedia.org/wikipedia/commons/thumb/2/28/Ethylene_glycol_chemical_structure.png/100px-Ethylene_glycol_chemical_structure.png"@@AMEPARAM@@commons/thumb/2/28/Ethylene_glycol_chemical_structure.png/100px-Ethylene_glycol_chemical_structure.png[/ame]*)
It's advisable to use Distilled water when changing coolant as tap water will create 'furring' in the cooling system, possibly blocking up smaller coolant galleries over time.
Concentrations vary depending on manufacturer and application but are generally between a 40%-60% mix. Altho Some coolants for colder climates may have a stronger mix of 70%-30% which can offer protection down to -64*C (-84*F) Make sure you check the bottle to ensure correct concentration for that particular coolant in your climate.
this is a rough guide to coolant concentrates and should be used for information only every coolant differs so make sure to read the bottle before starting.

*I cannot guarantee the accuracy of the information in this document nor do I take any responsibility for damage to any vehicle or person due to the misinterpretation of the information/inadequate skills, any work that you carry out to your own vehicle is entirely at your own risk. These are guidelines only and if in doubt I kindly suggest that you contact a Fiat dealer/relative organisation for clarification/legalities.*

